Nachwuchsgruppe Dr. Guido Wollmann
About | Projects | Team |Publications | Links
Oncolytic Virus Group
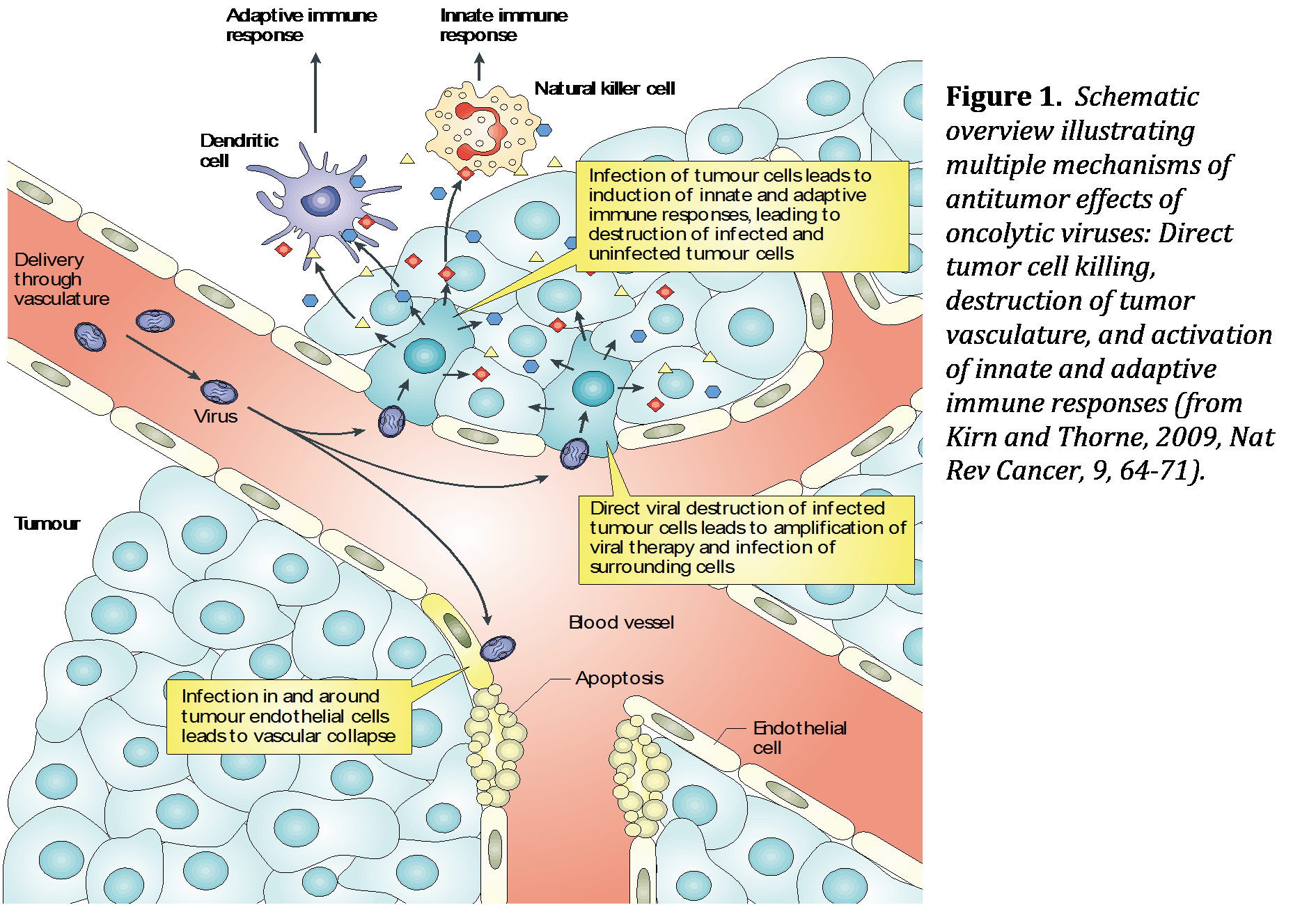
Our lab focuses on developing novel cancer therapeutics using oncolytic viruses. Oncolytic virotherapy is based on viruses with natural or engineered tumor tropism or tumor-selective replication. Tumor cell lysis leads to release of viral progeny, which go on to infect and kill neighboring tumor cells. Equally important to direct tumor cell lysis, however, is that the viral infection induces a local inflammation with subsequent immune cell infiltration. This can bring about the release of tumor-associated antigens, which are presented to T-cells by endogenous antigen presenting cells. In the nature of their design, compared to other pharmaceutical interventions, oncolytic viruses are able to amplify their therapeutic signals once delivered to the tumor. Modern oncolytic virus development started in the early 1990s with genetic modifications of herpes simplex viruses and adenoviruses. Since then, naturally occurring wild-type derivatives or genetically modified variants of about 20 viruses have been extensively tested in preclinical and clinical settings. In recent years, oncolytic virus development was boosted by significant advances made in the field of immunotherapy and strong indications for potential synergisms between the two. We are currently developing a novel oncolytic virus - VSV-GP - that shows a particular promise as a systemically applied virotherapy and stimulator of antitumoral immunity.
About VSV-GP as an oncolytic virus
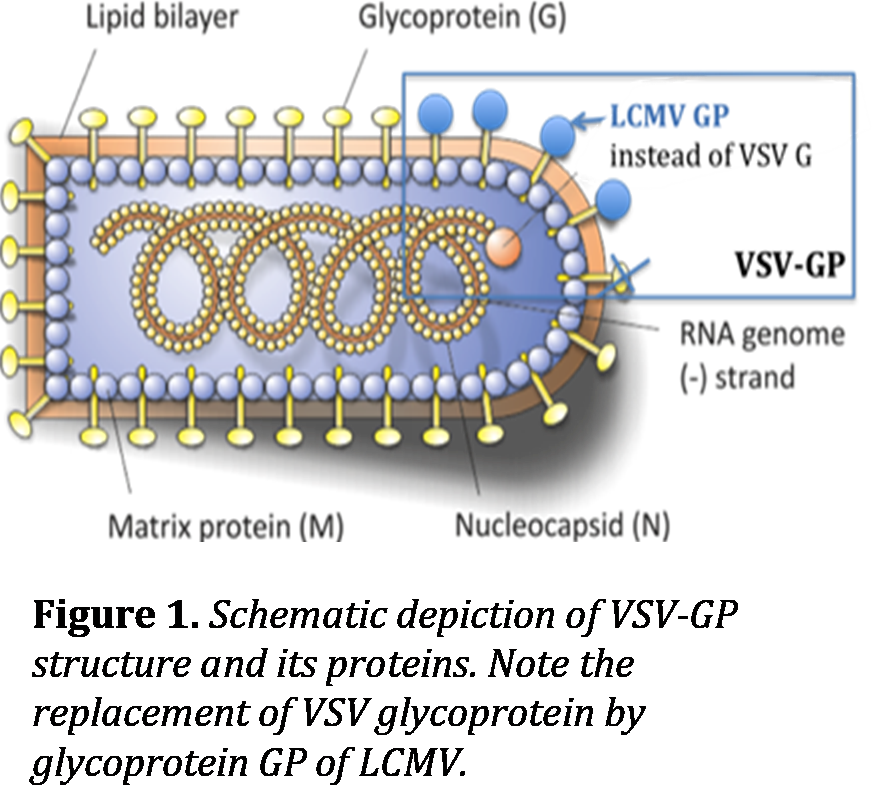
The oncolytic potential of vesicular stomatitis virus (VSV) was first described in the early 2000s. VSV is an enveloped RNA virus associated with mild and self-limiting disease in livestock. Human infection is very rare and usually asymptomatic. Consequently, most people don’t show preexisting immunity to VSV. The virus is very sensitive to interferon (IFN)-mediated antiviral control. Defects in the IFN pathway commonly found in cancer cells therefore form the base for VSV’s tumor selective replication. It is thus independent from specific tumor mutations and has been shown to target and kill a large variety of human cancer types. With its fast replication cycle, the lack of mutagenic potential to host cells, its capacity to deliver additional therapeutic genes and existing high titer production protocols, VSV has been one of the most promising oncolytic viruses. Still, a number of critical factors have limited its clinical development, namely the neurotoxicity of wildtype VSV and the rapid induction of neutralizing antibodies that prevent repeated intravenous applications. Dorothee von Laer and her oncolytic virus group overcame these limitations by generating a new chimeric virus, VSV-GP, in which the VSV “G” envelope protein is replaced by a foreign viral surface protein, the lymphocytic choriomeningitis virus (LCMV) glycoprotein “GP”, resulting in the chimeric replication competent virus “VSV-GP” (Figure 1). VSV-GP is non-neurotoxic, does not readily induce a protective antiviral antibody response and is stable in human serum (Muik et al., 2014). This forms the base for the projected clinical development of VSV-GP, which is spearheaded by ViraTherapeutics GmbH - a spin-off of the Medical University of Innsbruck. First-in-man clinical trials are expected to commence in the near future.
Research Projects
- Preclinical studies to prepare a first-in-man clinical trial with ViraTherapeutic’s VSV-GP
Studying tumor cell characteristics that support or inhibit VSV-GP activity
Tumor-stroma interaction with oncolytic viruses
Optimizing treatment regimen for a number of solid tumors, including prostate, ovarian, lung cancer and melanoma
Group Members
 Guido Wollmann, MD
Guido Wollmann, MD
Head of CD lab
Curriculum Vitae
 Sarah Danklmaier
Sarah Danklmaier
PhD-Student
 Jacqueline Schörgenhuber, MSc
Jacqueline Schörgenhuber, MSc
Technician
jacqueline.schörgenhuber@i-med.ac.at
 Nina Payr
Nina Payr
MSc Technician
Publications
Links:
http://www.ncbi.nlm.nih.gov/pubmed/
http://www.cancer.gov/about-cancer/causes-prevention/vaccines-fact-sheet