
Neuronal circuits underlying fear and anxiety
Neural circuits underlying fear and anxiety
Our main research interest is to understand the neural mechanisms mediating emotional information processing in the amygdala, and in particular how fear memories are formed, stored and supressed.
This work was and is supported by Austrian Science Funds (FWF) through single projects (P22969, P25375), the doctoral programme SPIN and the Special Research program SFB-F44.
In Project P22969, we have examined several structural and functional features of ITC neurons, a network of interconnected medium spiny GABAergic neurons organized in distinct clusters surrounding the BLA. Our work revealed an unexpected diversity in the pattern of their axonal projections that suggested the presence of distinct subpopulations (Busti et al., 2011). Moreover, we showed that these cells are an important site of convergence for glutamatergic sensory inputs. This finding has major relevance for our understanding of the formation of fear memories.
The encoding of fear memories is often studied both in rodents and in humans using classical Pavlovian fear conditioning. In fear conditioning, a neutral “conditioned” stimulus (CS, e.g. a tone) is paired with an aversive “unconditioned” stimulus (US, e.g. a mild footshock). Training leads to a CS-US association and to a conditioned fear response upon presentation of the CS alone. If the CS is repetitively presented in the absence of the US, a progressive decrease in the conditioned fear response is observed, a phenomenon, which is generally referred to as extinction learning. Extinction in animals is procedurally similar to forms of cognitive behavioral therapy that rely on exposure to anxiety-provoking cues in humans. Thus, fear extinction has considerable translational value and clinical relevance.
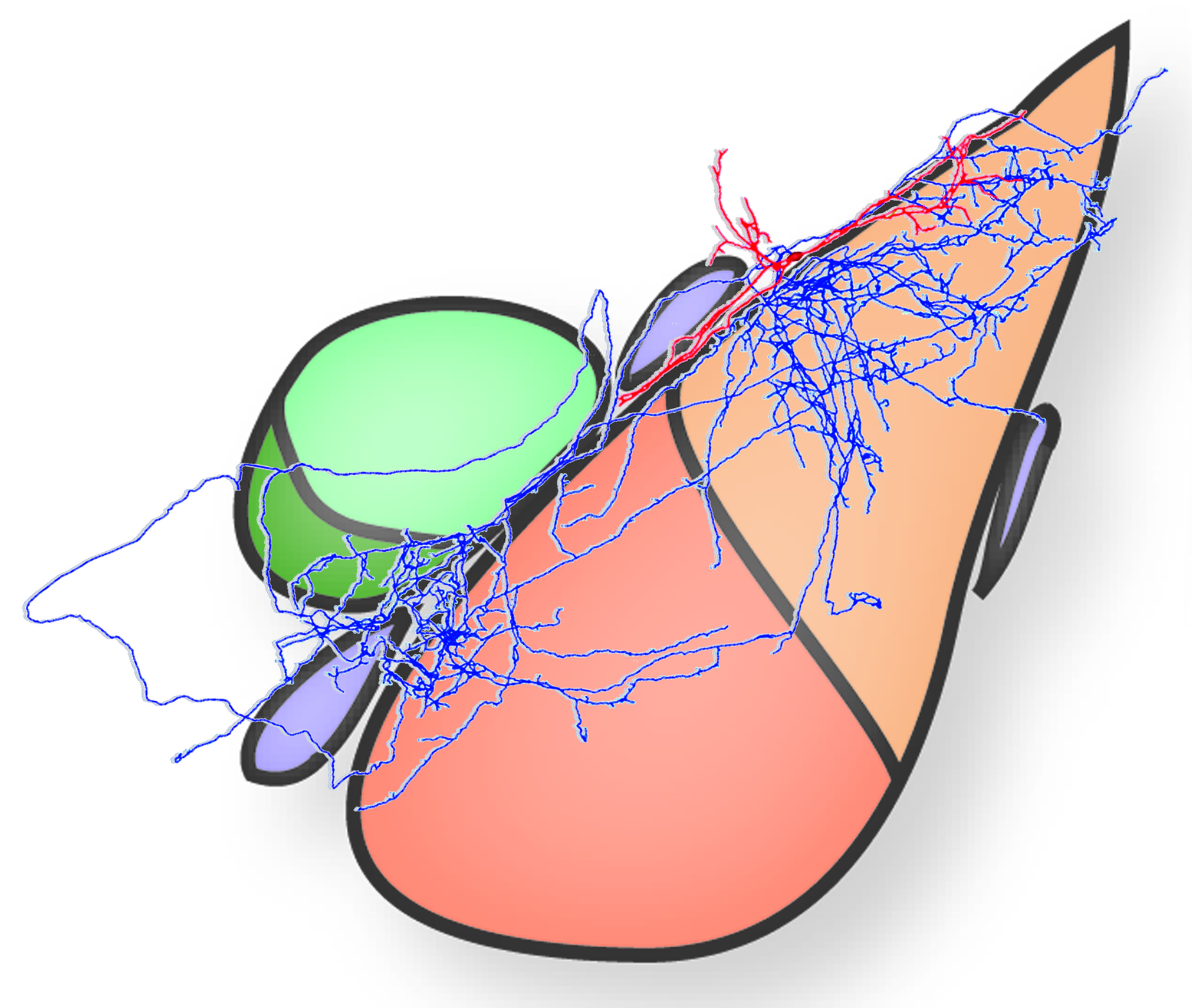
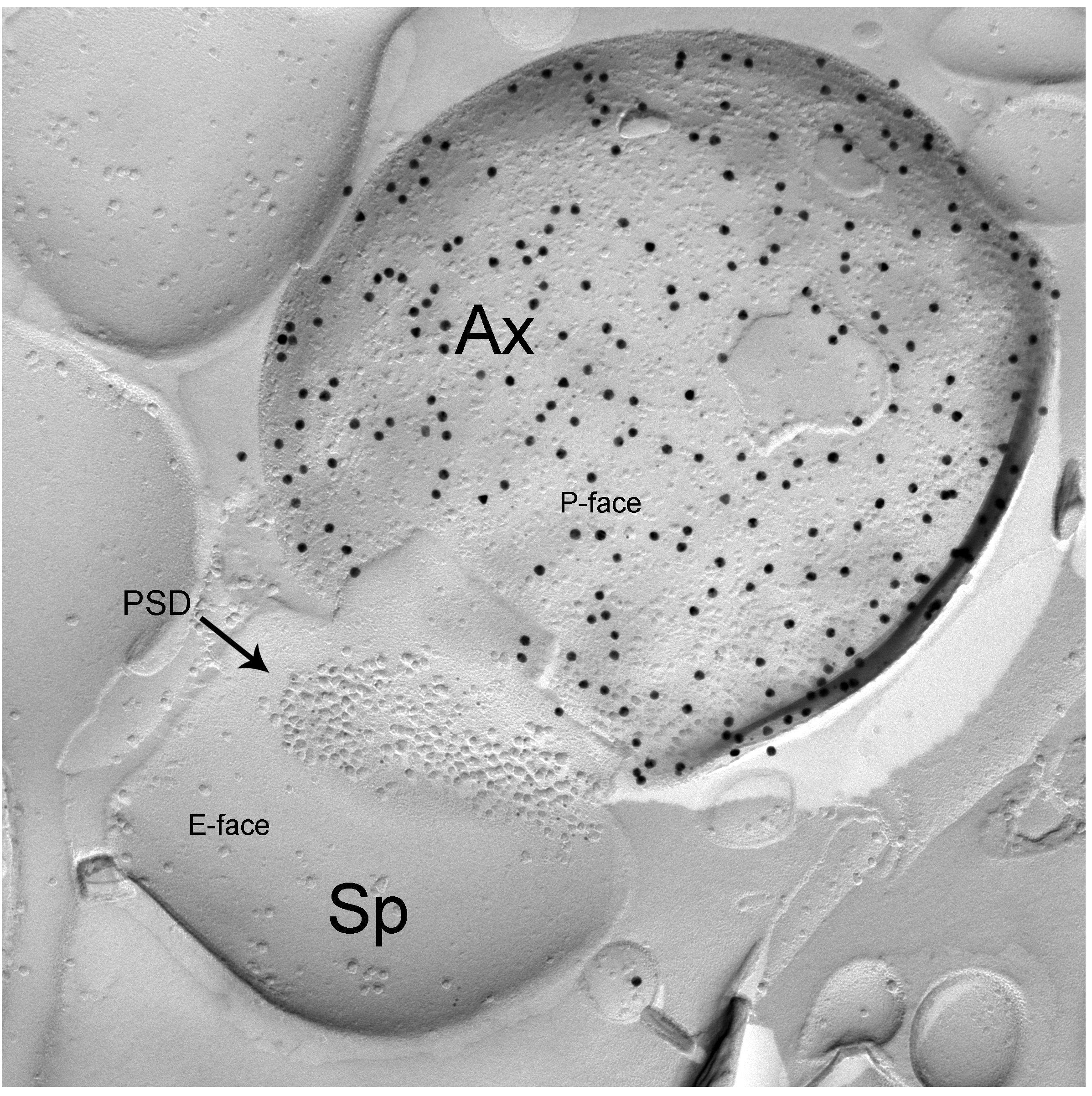
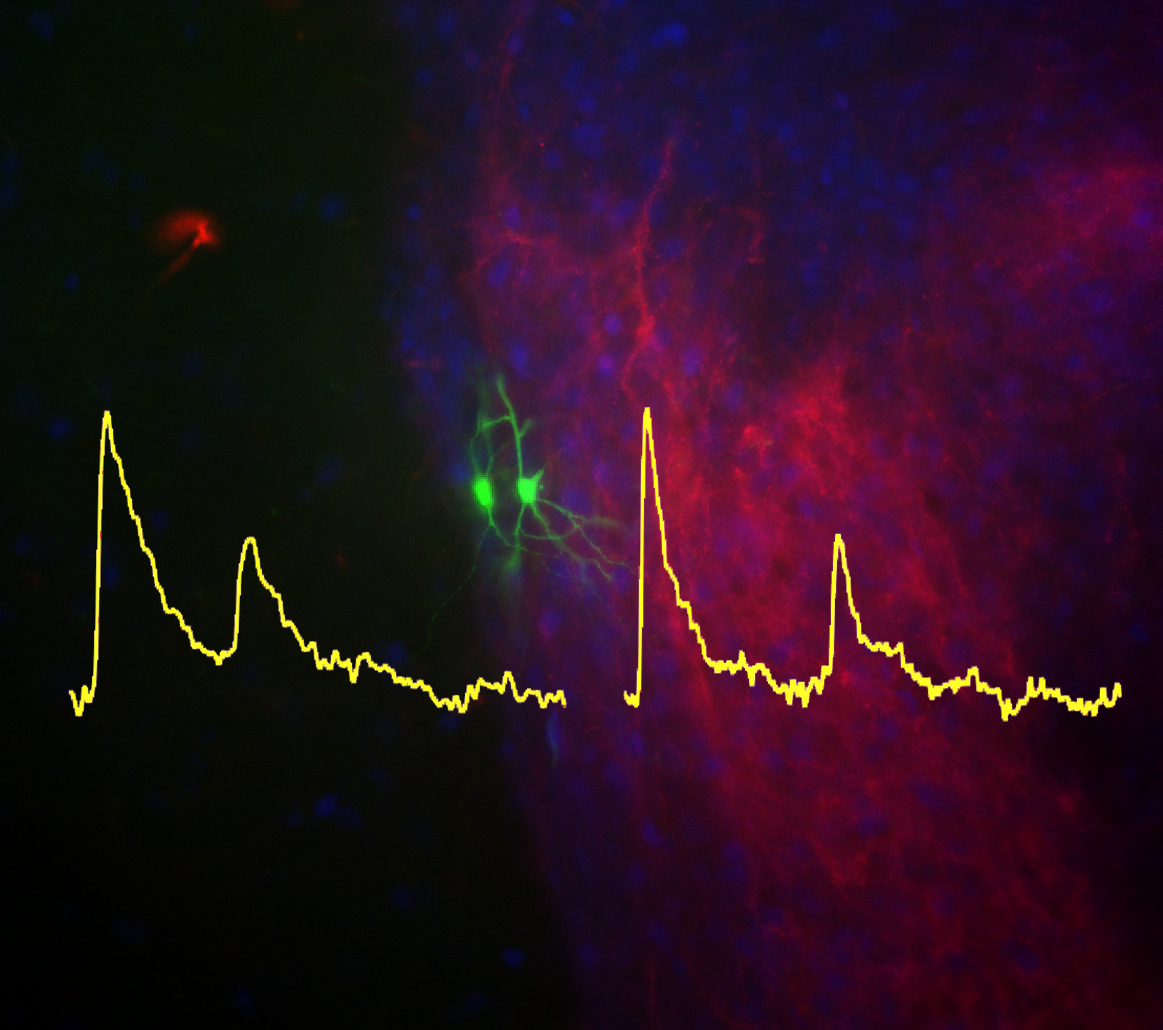
Taking advantage of recent developments in molecular genetics, viral trans-synaptic tracing and novel ultrastructural techniques (e.g. freeze-fracture replica immunogold labelling or FRIL), we investigate long-range connections between amygdala GABAergic neurons (e.g. interneurons of the BLA or ITC) and cortical or subcortical brain structures.
Moreover, we examine the pharmacological and anatomical bases of anxiety disorders in models of Parkinson`s disease. In particular, we seek to determine whether dopamine-depletion of the amygdala elicits pathological anxiety in mice.
Selected Publications:
Asede, D., Bosch, D., Luthi, A., Ferraguti, F., & Ehrlich, I. (2015). Sensory inputs to intercalated cells provide fear-learning modulated inhibition to the basolateral amygdala. Neuron, 86(2), 541-554.
Bienvenu, T. C., Busti, D., Micklem, B. R., Mansouri, M., Magill, P. J., Ferraguti, F., & Capogna, M. (2015). Large intercalated neurons of amygdala relay noxious sensory information. J Neurosci, 35(5), 2044-2057.
Kaufmann, W. A., Matsui, K., Jeromin, A., Nerbonne, J. M., & Ferraguti, F. (2013). Kv4.2 potassium channels segregate to extrasynaptic domains and influence intrasynaptic NMDA receptor NR2B subunit expression. Brain Struct Funct, 218(5), 1115-1132.
Bienvenu, T. C., Busti, D., Magill, P. J., Ferraguti, F., & Capogna, M. (2012). Cell-type-specific recruitment of amygdala interneurons to hippocampal theta rhythm and noxious stimuli in vivo. Neuron, 74(6), 1059-1074.
Sreepathi, H. K., & Ferraguti, F. (2012). Subpopulations of neurokinin 1 receptor-expressing neurons in the rat lateral amygdala display a differential pattern of innervation from distinct glutamatergic afferents. Neuroscience, 203, 59-77.
Busti, D., Geracitano, R., Whittle, N., Dalezios, Y., Manko, M., Kaufmann, W., . . . Ferraguti, F. (2011). Different fear states engage distinct networks within the intercalated cell clusters of the amygdala. J Neurosci, 31(13), 5131-5144.
Herry, C., Ferraguti, F., Singewald, N., Letzkus, J. J., Ehrlich, I., & Luthi, A. (2010). Neuronal circuits of fear extinction. Eur J Neurosci, 31(4), 599-612.
Metabotropic glutamate (mGlu) receptors in central nervous system function and dysfunction
Glutamate, like many other neurotransmitters, exerts its pleiotropic roles by means of multiple receptor proteins. Two main classes of glutamate receptors have been identified: ionotropic receptors, which are multimeric ion channels responsible for fast synaptic transmission, and metabotropic receptors, which couple to G-proteins to modulate slow synaptic transmission through intracellular second messengers. Metabotropic glutamate (mGlu1–8) receptors play a pivotal role in synaptic plasticity and many forms of learning and memory, and have received great interest as potential new drug targets for a broad range of diseases including schizophrenia, autism, general anxiety disorder and even cancer.
Using a number of different approaches ranging from proteomic to immune-electron microscopy, we aim to further delineate the precise subcellular distribution of these receptors in distinct brain areas as well as their participation to macromolecular complexes regulating intracellular signalling.
Project I-2220
In a joint project with the Aiba group (University of Tokyo) we will investigate the functional significance of mGlu1 receptor splice variants. Four translated alternatively spliced variants of the mGlu1 receptor have been identified, which are characterized by a common N-terminal extracellular domain but different C-termini of variable length. For our studies, we will take advantage of the availability of different genetically modified mouse lines exclusively expressing the mGlu1a or mGlu1b in cerebellar Purkinje cells. Novel mouse lines will also be developed to address the role and localization of the mGlu1g isoform.
Selected Publications:
Mansouri, M., Kasugai, Y., Fukazawa, Y., Bertaso, F., Raynaud, F., Perroy, J., . . . Ferraguti, F. (2015). Distinct subsynaptic localization of type 1 metabotropic glutamate receptors at glutamatergic and GABAergic synapses in the rodent cerebellar cortex. Eur J Neurosci, 41(2), 157-167.
Dobi, A., Sartori, S. B., Busti, D., Van der Putten, H., Singewald, N., Shigemoto, R., & Ferraguti, F. (2013). Neural substrates for the distinct effects of presynaptic group III metabotropic glutamate receptors on extinction of contextual fear conditioning in mice. Neuropharmacology, 66, 274-289.
Nicoletti, F., Bockaert, J., Collingridge, G. L., Conn, P. J., Ferraguti, F., Schoepp, D. D., . . . Pin, J. P. (2011). Metabotropic glutamate receptors: from the workbench to the bedside. Neuropharmacology, 60(7-8), 1017-1041.
Ferraguti F., Crepaldi L. and Nicoletti F. (2008) Metabotropic glutamate receptor 1 (mGlu1): Current concepts and perspectives. Pharmacol Rev, 60:536-581.
Other Recent Publications
Lange, M. D., Jungling, K., Paulukat, L., Vieler, M., Gaburro, S., Sosulina, L., Blaesse P., Sreepathi H.K., Ferraguti F., Pape, H. C. (2014). Glutamic acid decarboxylase 65: a link between GABAergic synaptic plasticity in the lateral amygdala and conditioned fear generalization. Neuropsychopharmacology, 39(9), 2211-2220.
Brunner, J., Ster, J., Van-Weert, S., Andrasi, T., Neubrandt, M., Corti, C., Corsi M., Ferraguti F., Gerber U. and Szabadics, J. (2013). Selective silencing of individual dendritic branches by an mGlu2-activated potassium conductance in dentate gyrus granule cells. J Neurosci, 33(17), 7285-7298.
Whittle, N., Schmuckermair, C., Gunduz Cinar, O., Hauschild, M., Ferraguti, F., Holmes, A., & Singewald, N. (2013). Deep brain stimulation, histone deacetylase inhibitors and glutamatergic drugs rescue resistance to fear extinction in a genetic mouse model. Neuropharmacology, 64, 414-423.
National Collaborators
Singewald Nicolas, Pharmacology & Toxicology, University of Innsbruck
Lindner Herbert, Section of Clinical Biochemistry, Medical University of Innsbruck
International Collaborators
Aiba Atsu, The University of Tokyo, Tokyo
Capogna Marco, Oxford University
Ehrlich Ingrid, Hertie-Institute, University of Tübingen
Fisone Gilberto, Karolinska Institute, Stockholm
Lüthi Andreas, Friedrich Miescher Institute, Basel
Alumni
Luca Crepaldi, Resarch Fellow, MRC/University College London, London, UK
Carmen Lackner, Lecturer, Management Centre Innsbruck, Innsbruck, Austria
Daniela Busti, Lecturer, Verona, Italy
Institut für Pharmakologie
Kontakt:
Iris Markt
Tel.: +43 (0)512/9003-71201
E-Mail: iris.markt@i-med.ac.at
Fax: +43 (0)512/9003-73200
E-Mail: pharmakologie@i-med.ac.at
Peter-Mayr-Straße 1a
A-6020 Innbruck
Sie finden uns hier.
research funded by:



Group Leader
Postdoc
Postdoc
PostDoc
PhD Student
Lab Assistant
Master Students:
- Luisa-Marie Zäuner
- Alice Vincenzi
- Stefano Pioli









