 ResearchGate
ResearchGate
 Google Scholar
Google Scholar
Renal Epithelial Physiology and Toxicology

From left to right:
Alice Limonciel, Giada Carta, Paul Jennings and Anja Wilmes. (Picture was taken in January 2016 at the System Toxicology Conference in Les Diablerets, Switzerland).
Our research: The kidney and susceptibility to injury
Each kidney consists of approximately 1 million nephrons at birth. De novo nephrogenesis does not occur after birth and there is no strong evidence suggesting that the adult kidney harbours resident stem cells. Thus, the kidney unlike the liver is not a highly rejuvenative organ, which is most likely an evolutionary compromise allowing the anatomical complexity required for high function and the maintenance of the very narrow margins required for whole body homeostasis. Indeed, the kidney is an extremely accomplished organ and can carry out 100 % of its duties with only a fraction of the nephrons we are born with. However, we continually lose nephrons through-out life and will, all things being equal, eventually breach the renal functional reserve and enter end-stage-renal disease. Thus, anything that contributes to chronic renal failure has the potential to seriously curtail life quality and life-span. Aging populations and associated risk factors such as diabetes and heart disease, have pushed chronic kidney disease (CKD) incidence to unprecedented levels (currently at 10 % of the European population). Due to the role of the kidney in elimination of waste products, renal cells, particularly the proximal tubule, will internally process the majority of drugs and chemicals, and thus often have higher concentrations of these compounds than any other cell in the body. Compounds that injure renal epithelial cells can initiate and/or accelerate CKD.
Latest publications:
2016: Inter-laboratory study of human in vitro toxicogenomics-based tests as alternative methods for evaluating chemical carcinogenicity: a bioinformatics perspective. Herwig, R., Gmuender, H., Corvi, R., Bloch, K.M., Brandenburg, A., Castell, J., Ceelen, L., Chesne, C., Doktorova, T.Y., Jennen, D., Jennings, P., Limonciel, A., Lock, E.A., McMorrow, T., Phrakonkham, P., Radford, R., Slattery, C., Stierum, R., Vilardell, M., Wittenberger, T., Yildirimman, R., Ryan, M., Rogiers, V., and Kleinjans, J. Archives of toxicology 90, 2215-2229 https://www.ncbi.nlm.nih.gov/pubmed/26525393
2016: Development and characterization of a pseudo multiple reaction monitoring method for the quantification of human uromodulin in urine. Hammond, T.G., Moes, S., Youhanna, S., Jennings, P., Devuyst, O., Odermatt, A., and Jeno, P. Bioanalysis 8, 1279-1296 http://www.ncbi.nlm.nih.gov/pubmed/27187191
2016: „Watching the Detectives“ report of the general assembly of the EU project DETECTIVE Brussels, 24-25 November 2015. Fernando, R.N., Chaudhari, U., Escher, S.E., Hengstler, J.G., Hescheler, J., Jennings, P., Keun, H.C., Kleinjans, J.C., Kolde, R., Kollipara, L., Kopp-Schneider, A., Limonciel, A., Nemade, H., Nguemo, F., Peterson, H., Prieto, P., Rodrigues, R.M., Sachinidis, A., Schafer, C., Sickmann, A., Spitkovsky, D., Stober, R., van Breda, S.G., van de Water, B., Vivier, M., Zahedi, R.P., Vinken, M., and Rogiers, V. Archives of toxicology 90, 1529-1539 http://www.ncbi.nlm.nih.gov/pubmed/27129694
2016: Improving global feature detectabilities through scan range splitting for untargeted metabolomics by high-performance liquid chromatography-Orbitrap mass spectrometry. Ranninger, C., Schmidt, L.E., Rurik, M., Limonciel, A., Jennings, P., Kohlbacher, O., and Huber, C.G. Anal Chim Acta 930, 13-22 http://www.ncbi.nlm.nih.gov/pubmed/27265900
2016: Moving Beyond Prioritization Toward True In Vitro Safety Assessment. Yoon, M., Adeleye, Y., Clewell, R., Jennings, P., and Whelan, M. Applied In Vitro Toxicology 2, 67-73 http://dx.doi.org/10.1089/aivt.2016.29005.rtl
2016: Ensuring the Quality of Stem Cell-Derived In Vitro Models for Toxicity Testing. Stacey, G.N., Coecke, S., Price, A.B., Healy, L., Jennings, P., Wilmes, A., Pinset, C., Ingelman-Sundberg, M., Louisse, J., Haupt, S., Kidd, D., Robitski, A., Jahnke, H.G., Lemaitre, G., and Myatt, G. Advances in experimental medicine and biology 856, 259-297 http://www.ncbi.nlm.nih.gov/pubmed/27671727
2015: Mechanism of cisplatin proximal tubule toxicity revealed by integrating transcriptomics, proteomics, metabolomics and biokinetics. Wilmes, A., Bielow, C., Ranninger, C., Bellwon, P., Aschauer, L., Limonciel, A., Chassaigne, H., Kristl, T., Aiche, S., Huber, C.G., Guillou, C., Hewitt, P., Leonard, M.O., Dekant, W., Bois, F., and Jennings, P. Toxicology in vitro : an international journal published in association with BIBRA 30, 117-127 http://www.ncbi.nlm.nih.gov/pubmed/25450742
Congratulations to Anja for winning the 1st Otto-Kraupp-Preis 2017

Paul’s Henry Stewart Talk contribution
Jennings, P. (2016), „Molecular basis of genetic renal diseases 1 and 2“, in The Biomedical & Life Sciences Collection, Henry Stewart Talks Ltd, London (online at https://hstalks.com/bs/3413/, https://hstalks.com/bs/3414/ )
Anja collects her 2016 TWF award for her proposal on claudin regulation in oxidative stress.
https://www.i-med.ac.at/mypoint/news/697115.html
Alice wins the CEFIC LRI award 2015 !
https://www.i-med.ac.at/mypoint/news/696075.html
And the follow up:
Highlighted Articles
Evidence for a role of claudin 2 as a proximal tubular stress responsive paracellular water channel.
Published in Toxiciology and Applied Pharmacology, September 2014.
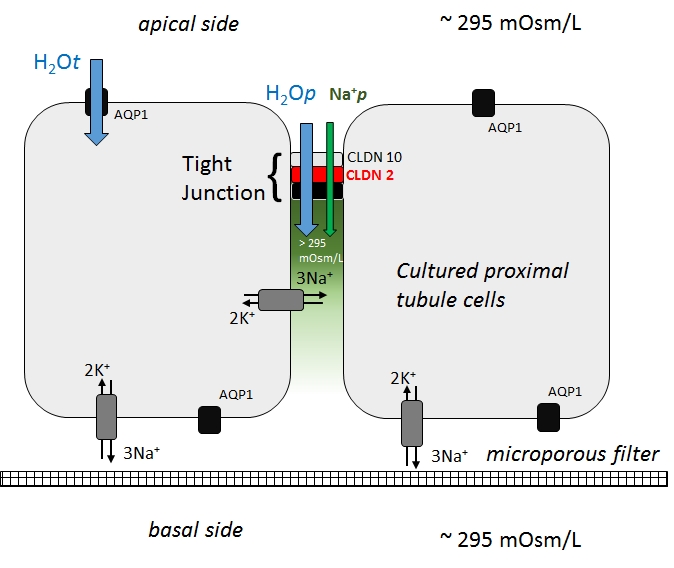 |
How water passes through the paracellular space is somewhat of a mystery. In our investigations with nephrotoxic compounds in cultured renal epithelial cells we have discovered that some compounds cause the selective decrease in the expression of specific tight junction proteins for example claudin 2 and claudin 10. In claudin 2 knock down cells we observed an inability of the cells to reabsorb water, but only when cultured on microporous filters. Our explanation is that an osmotic gradient in the paracellular space drives water through tight junctions, but only if claudin 2 is present. Thus claudin 2 is a putative paracellular water channel. Download article here. |
Application of integrated transcriptomic, proteomic and metabolomic profiling for the delineation of mechanisms of drug induced cell stress
Published in the Journal of Proteomics in January 2013.
|
|
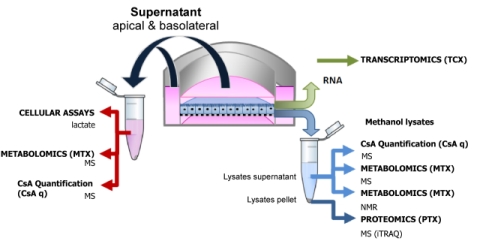
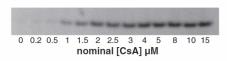
In this paper we investigate the effects of therapeutic and supra theraputic concentrations of the immunosupressive compound Cyclosporine A (CsA) on cultured human renal proximal tubule cells. We utilised a high content non-biased omic strategy to identify altered transcripts, proteins and metabolites. In addition, together with our collegues in Würtzburg University and Université de Technologie de Compiègne we developed a kinetic model of CsA cell uptake. The paper is the first of its kind to integrate omic technologies to identify compound induced cell stress. It is likely that pharmacological and toxicological screening in the future will employ similar strategies of human cell based systems, integrated omics and biokinetics. Download full open access article here. |
Our Book Contributions
 |
 |
 |
 |
Our research in the public domain
 |
Left: Alice Limonicel receives the Lush young scientist prize in London, 2013. |
Projects
Horizon 2020, EU-ToxRisk, „An Integrated European ‘Flagship’ Program Driving Mechanism-based Toxicity Testing and Risk Assessment for the 21st Century„
EU 7th Framework and EFPIA, StemBANCC „Stem cells for Biological Assays of Novel drugs and predictive toxiCology „
EU 7th Framework and Cosmetics Europe, DETECTIVE „Detection of endpoints and biomarkers for repeated dose toxicity using in vitro systems „
Recently Completed Projects
The NC3R Nephrotube – „Development of a multi-compartmental, microfluidic tissue model of tubular injury in assays of nephrotoxicity.“
EU 7th Framework, Predict-IV „Profiling the toxicity of new drugs: a non animal-based approach integrating toxico-dynamics and biokinetics „
EU 6th Framework, carcinoGENOMICS „Novel alternative method approaches in carcinogenicity and mutagenicity.“