Molecular Biology
Research Focus
General Facts
Research
Selected Publications
Selected Funding, Collaboration
Keywords: Chromatin and epigenetics, RNA cytosine methylation, filamentous fungi, iron metabolism, fungal infection, drug resistance, bioactive fungal products, siderophores, lipocalins, antimicrobial proteins
Research (ÖSTAT Classification) : 106023, 106022, 106024, 301114, 303020
Research Focus
Fungal Metabolism, Gene Regulation, Virulence and Drug Resistance
- Iron metabolism in filamentous fungi: links to human disease
- Histone-modifying enzymes in gene regulation, in fungal physiology and as antifungal targets
- Structure, mechanism of action and applicability of secreted antimicrobial peptides/proteins
- Antifungal therapeutics and resistance
Epigenetics and Epitranscriptomics
- Chromatin assembly mechanisms: Chromatin remodellers and histone variants
- RNA methylation and its impact on RNA metabolism
General Facts
The Institute of Molecular Biology is home to five independent research groups, whose scientific interests range from investigating diverse aspects of filamentous fungal physiology and metabolism to studying secretory lipocalins and researching the nature and significance of chromatin remodelling and epitranscriptomic mechanisms. The aim of all research groups is ultimately to translate scientific discoveries into the diagnosis and treatment of human disease. Continuous third-party funding (e.g. FWF) of all research groups and the participation of members of the institute in various intra and extramural network activities, such as the FWF-funded PhD programmes “HOROS” and “CBD”, the Euregio programme “SupErA” and SFB “RNA Deco”, are a testament to the high standard of research. Staff members at the Institute of Molecular Biology also contribute substantially to curricular teaching and administration activities at MUI. Notably, former institute chair (until 2019) Peter Loidl is Vice Rector for Academic Affairs at the university. Bachelors’ and masters’ courses in molecular medicine are coordinated by Gerald Brosch, who succeeded Bernhard Redl (retired in 2019) in the role. Moreover, Gerald Brosch also organises the “Genetics, Epigenetics and Genomics” PhD programme. Beyond this, all group leaders teach lectures, seminars and practical courses as part of the human medicine, dental medicine, molecular medicine (bachelors’ and masters’) and PhD curricula.
Research
Iron Metabolism of Filamentous Fungi: Links to Human Disease
Hubertus Haas
Aspergillus fumigatus is a typical saprobic mould fungus but also the most common human airborne fungal pathogen in Europe. Owing to unsatisfactory diagnostic and therapeutic options, it causes allergic and life-threatening invasive diseases, depending on the immune status of the patient. Our research goal is the characterisation of fungal metabolism, in order to improve antifungal therapy, the diagnosis of fungal infections and the biotechnological potential of filamentous fungi. The present research focuses on iron homeostasis-maintaining mechanisms of Aspergilli. We have demonstrated that siderophore-mediated iron acquisition and the iron-sensing transcription factor HapX are essential for virulence of A. fumigatus in murine aspergillosis models. Consequently, fungal iron metabolism represents an attractive target for the improvement of antifungal therapy and the diagnosis of fungal infections.
Major Achievements
Characterisation of siderophore-mediated iron acquisition and storage. Characterisation of fungal mechanisms for iron sensing and regulation. Metabolic linking of iron regulation, e.g. to ergosterol biosynthesis, hypoxia adaptation and leucine biosynthesis. In vivo PET (positron emission tomography) imaging of fungal infections using 68Gallium-labelled siderophores in animal models (Fig. 1, collaboration with Dr. Clemens Decristoforo, Department of Nuclear Medicine). Proof of concept for siderophores (triacetylfusarinine C) as urine biomarkers for aspergillosis.
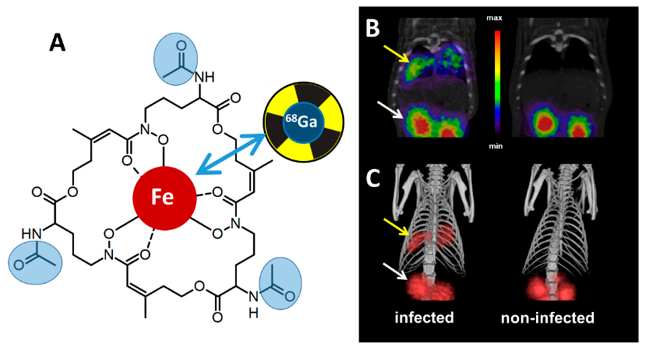
Fig. 1: Siderophore-mediated PET (positron emission tomography) imaging of aspergillosis with 68gallium-labelled triacetylfusarinine C (A) µPET/CT coronal slices (B) and volume-rendered 3D images (C) of A. fumigatus in a rat infection model, showing accumulation in infected lung tissue (yellow arrows) and accumulation in kidneys (white arrows) caused by renal excretion of the siderophore (Petrik et al., J. Fungi (Basel) 2020).
Future Goals
Further characterisation of fungal iron homeostasis and medical translation. Role of iron and siderophores in microbial interactions.
Functions of Histone-Modifying Enzymes in Gene Regulation and Fungal Physiology
Gerald Brosch and Stefan Graessle
In addition to distinct regulatory sequences in gene promoters, the readout of genetic information in eukaryotes is significantly controlled at the chromatin level. For instance, covalent post-translational modifications (PTMs) of histones and other proteins have profound structural and functional consequences for the transcriptional programme of a cell. Our research focuses on elucidation of the functional impact of lysine acetylation and arginine methylation on fungal physiology. In particular, we are interested in studying the extent to which modifying activities are involved in fungal pathogenicity as well as investigating their role in regulating secondary metabolite production. To this end, the group has generated Aspergillus strains with individual or multiple deletion respectively of all protein arginine methyltransferase (PRMT) genes and all classical lysine deacetylase (KDAC) genes. Using these tools together with specifically engineered transgenes, the impact on the viability, pathogenicity and metabolism of Aspergilli is studied. Moreover, we employ proteomic and transcriptomic analyses: (i) to characterise novel substrates and (ii) to investigate the contribution of KDACs and PRMTs to the regulation of secondary metabolism as well as stress response.
Major Achievements
KDACs: Identification and functional characterisation of two fungus-specific protein domains in the KDAC RpdA, which are essential for the viability of Aspergillus and may therefore serve as targets in novel antifungal therapies. Confirmation of RpdA as an essential fungal virulence factor in a mouse model for invasive pulmonary aspergillosis. Identification of a novel fungus-specific RpdA complex that is essential for sexual reproduction (Fig. 2). Proof that KDAC HosA is a major regulator of medically important secondary metabolites of filamentous fungi.
PRMTs: Functional linking of Aspergillus type I and II PRMTs to various cellular processes, including detoxification, secondary metabolism, transport and protein secretion. Identification of novel PRMT substrates involved in RNA metabolism.
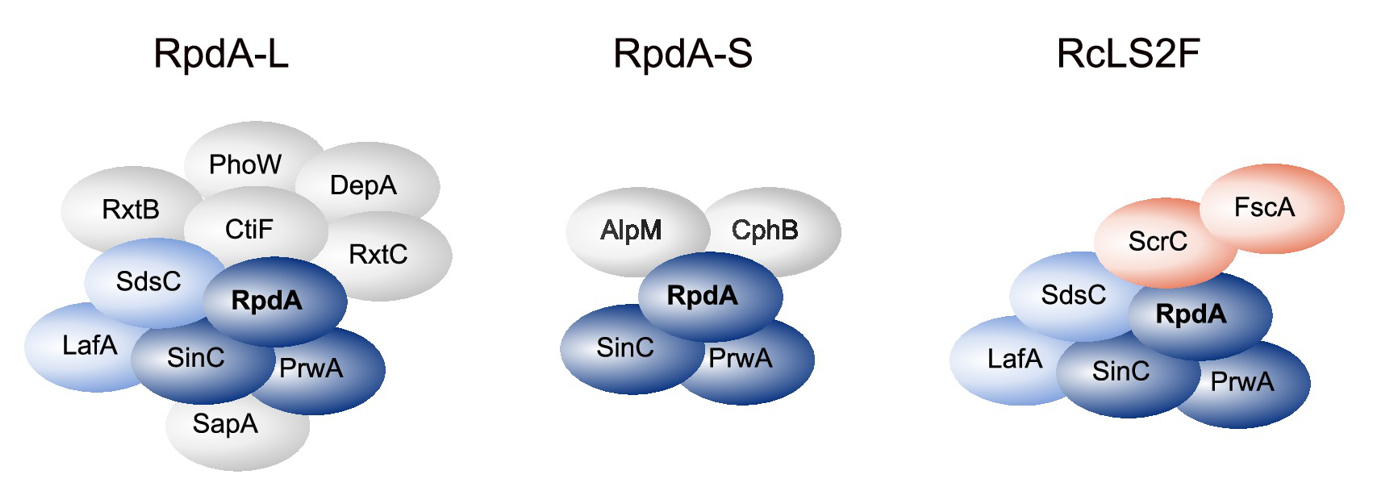
Fig. 2: RpdA/SinA complexes including the novel RcLS2F complex in Aspergillus nidulans. (Bauer et al., Front. Microbiol. 2020)
Future Goals
KDACs: Screening for new KDAC inhibitors with potential as antifungals, preferably targeting RpdA-specific domains. Identification of (novel) KDAC-regulated secondary metabolites (SM) of Aspergilli and elucidation of the control mechanisms of the corresponding SM gene clusters via KDACs. Further identification of components of different KDAC complexes and their significance for the biological function of these KDACs.
PRMTs: Isolation and characterisation of methylated proteins in stress-induced cells and along specific metabolic processes. Analysis of regulatory complexes to assess PRMT methylation activity. Elucidation of the biological role of as yet uncharacterised type IV PRMTs.
Structure, Mechanism of Action and Applicability of Antimicrobial Peptides / Proteins Secreted by Filamentous Fungi
Florentine Marx-Ladurner
Filamentous fungi secrete a wide range of different proteins into the external medium, which are used for various functions, such as nutrient assimilation, quorum sensing, host invasion and colonisation etc. Apart from some secreted enzymes that have been developed for a variety of commercial uses (mainly for the fermentation industry), few extracellular proteins are characterised well with respect to function, such as pathogenicity or cell signalling factors. Our main scientific interest is in identification, isolation and further characterisation at the molecular, structural and functional level of novel extracellular proteins with antimicrobial activity from filamentous ascomycetes, e.g. Penicillium chrysogenum, Aspergillus nidulans and Aspergillus fumigatus (Fig. 3). Antimicrobial proteins are promising candidates for the development of novel therapies applicable in medicine as well as in agriculture and the food industry for the prevention and treatment of microbial infections. Detailed characterisation of these proteins is therefore of crucial importance and essential for the development of new therapeutic approaches and their successful application in the future.
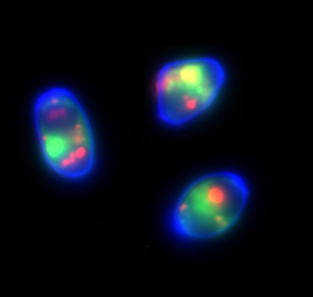
Fig. 3: Live-cell imaging of Neurospora crassa conidia exposed to BODIPY-labelled antifungal protein PAF (green, vacuoles) and co-stained with propidium iodide (red, nucleic acids) and calcofluor-white (blue, fungal cell wall).
Major Achievements
Understanding the structure-function relationship of antimicrobial proteins and first steps towards their biotechnological/medical application.
Future Goals
Identification of molecular targets for the development of new therapeutic drugs. Rational design of antimicrobial proteins and peptides with improved efficacy and specificity. Characterisation of additional functions of antimicrobial proteins in their host, in addition to their antimicrobial activity.
Antifungal Therapeutics and Resistance
Fabio Gsaller
As fungi belong to the domain of eukaryotes, many vital processes in mammalian cells are conserved in fungal species. Consequently, substances with antifungal activity often have toxic effects on human cells, which makes the development of novel antifungal drugs particularly challenging. In the clinical setting, the treatment of choice for Aspergillus infections is azole-based antifungals. However, emerging resistance to this drug class presents a growing threat to human health.
Our research explores metabolic effects caused by antifungal drugs as well as molecular factors employed by Aspergillus fumigatus to protect itself from antimycotic substances. In this regard, major aspects of our work involve mode-of-action determination and the investigation of gene regulatory mechanisms that drive resistance to clinically administered antimycotics, which is critical to the development of alternative strategies for the treatment of fungal infections.
Major Achievements
Elucidation of the molecular cause underlying one of the predominant resistance mechanisms found in A. fumigatus clinical isolates that leads to treatment failure. Discovery of the gene regulatory mechanism that confers intrinsic resistance on antifungal nucleobase analogue 5-fluorocytosine (Fig. 4).
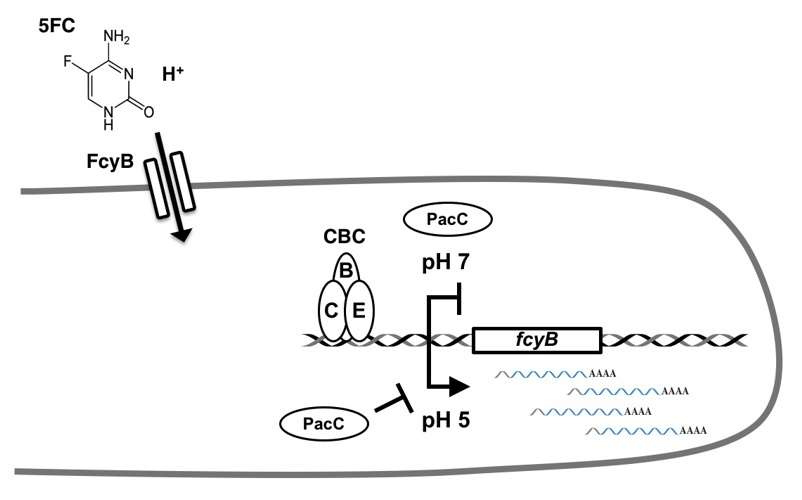
Fig. 4: Model illustrating pH-dependent regulation of 5-fluorocytosine (5FC) uptake through the CCAAT binding complex (CBC) and the pH regulator PacC. At physiological pH, both regulators repress the expression of the gene fcyB, which encodes for the major 5FC uptake protein in A. fumigatus. This repression is responsible for the intrinsic resistance of this mould pathogen to 5FC (Gsaller et al., Antimicrob. Agents Chemother 2018).
Future Goals
To increase our knowledge of strategies employed by fungal pathogens to counteract the activity of antifungal drugs, with the ultimate aim of improving current approaches to the treatment of fungal infections.
Lipocalins and their Involvement in Innate Immunity and Allergy
Bernhard Redl
We investigate structural and functional features of human lipocalins. The lipocalin superfamily consists of small, mainly secretory proteins defined on the basis of conserved amino acid sequence motifs and their common structure. Functionally, these are important extracellular carriers of lipophilic compounds in vertebrates, invertebrates, plants and bacteria. There is increasing evidence that this group of proteins is involved in a variety of physiological processes, including retinoid, fatty acid and pheromone signalling, immunomodulation, inflammation, detoxification, modulation of growth and metabolism, tissue development, apoptosis, and even behavioural processes. Whereas the structural basis of lipocalin-ligand binding is now well-understood, there is a major lack of knowledge with regard to the mechanisms by which lipocalins exert their biological effects. This is mainly due to the fact that only limited data are available on lipocalin receptors and lipocalin receptor interactions, although it is well accepted that many, if not all, of these proteins are able to bind to specific cell receptors. Our main research focus is on the identification of cellular lipocalin receptors, the characterisation of the molecular mechanisms of receptor-ligand interaction and the biological processes beyond receptor binding (Fig. 5). In addition, we study novel functions of lipocalins in innate immunity and allergy.
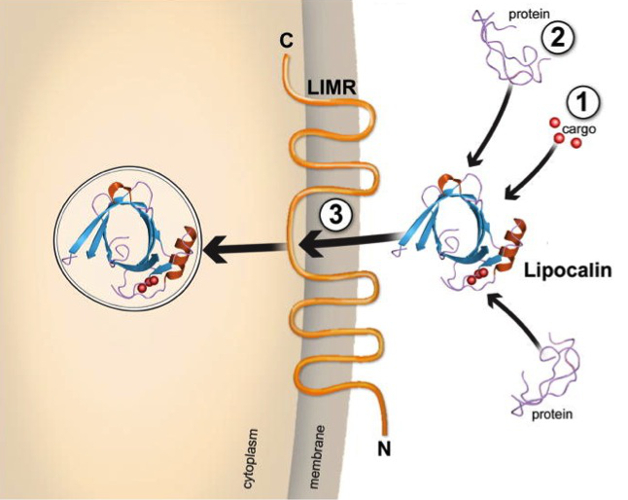
Fig. 5: Types of molecular recognition properties of lipocalins. (1) Lipophilic ligands illustrated as cargo. (2) Soluble macromolecule ligands, such as proteins. (3) A lipocalin-specific membrane receptor is responsible for cellular uptake of the lipocalin-ligand complex.
Major Achievements
Isolation of two novel lipocalin receptor candidates by membrane protein cross-linking and phage display.
Epigenetics and Epitranscriptomics
Alexandra Lusser
Eukaryotic DNA is assembled into a nucleoprotein complex called chromatin, of which the basic repeating unit is the nucleosome. The way in which DNA is organised in chromatin allows highly efficient compaction of the genetic material and provides additional levels of control in the regulation of nuclear processes, such as transcription, replication, repair and recombination. We are interested in learning how the establishment and maintenance of eukaryotic chromatin affects these processes. We are approaching this question by studying the molecular mechanisms and biological context of chromatin assembly and remodelling processes. Major research questions in my laboratory are: (i) the study of biological functions of ATP-dependent chromatin-remodelling factor CHD1 in Drosophila and in mice, (ii) the study of centromeric chromatin assembly in Drosophila and (iii) the analysis of 5-methylcytosine distribution and function in mammalian mRNA (“epitranscriptomics”).
Major Achievements
Recent results include the finding that the restricted incorporation of histone H3 variant CENP-A into centromeric chromatin as well as its maintenance in chromatin are strongly regulated by specific post-translational modifications in the N-terminus of CENP-A. Our studies on CHD1 have shown that this molecular motor plays an important role in learning and memory in mice, by controlling the expression of immediate early genes in the hippocampus. With respect to the epitranscriptomics project, we reported the first transcriptome-wide map of 5-methylcytosine in the poly(A)RNA of mice. Finally, in collaboration with Ronald Micura’s laboratory (University of Innsbruck), we developed TUC-seq and TUC-seq DUAL, two methods that use a particular conversion chemistry to allow the detection of metabolically labelled (nascent) RNA by means of sequencing-based techniques, in order to facilitate the study of RNA expression dynamics (Fig. 6).
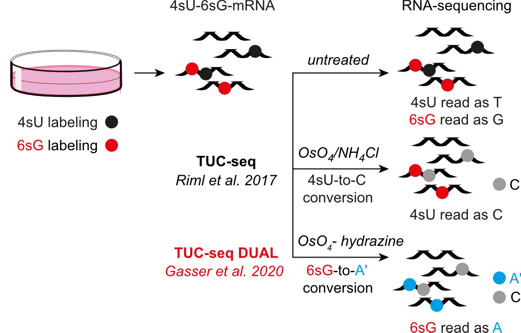
Fig. 6: Basic concept of TUC-seq and TUC-seq DUAL. RNA expression dynamics can be studied by means of metabolic labelling of nascent RNAs with 4sU and/or 6sG. The quantification of newly synthesised versus existing RNA is achieved by means of chemical conversion treatment of the isolated RNA followed by deep sequencing. RNAs that contain the labelling nucleotide are detected from the presence of U-to-C (in case of 4sU) and G-to-A (in case of 6sG) mutations (Gasser et al. Angew. Chem. Int. Ed. 2020).
Future Goals
Future studies will further explore chromatin assembly mechanisms of CENP-A, examine the role of CHD1 in Drosophila metabolism and aging, and continue to elucidate the physiological significance of RNA cytosine modifications.
Selected Publications
- Hoenigl, Martin; Orasch, Thomas; Faserl, Klaus; Prattes, Juergen; Loeffler, Juergen; Springer, Jan; Gsaller, Fabio; Reischies, Frederike; Duettmann, Wiebke; Raggam, Reinhard B.; Lindner, Herbert; Haas, Hubertus: Triacetylfusarinine C: A urine biomarker for diagnosis of invasive aspergillosis. JOURNAL OF INFECTION. 2019; 78(2); 150-157
- Misslinger, Matthias; Scheven, Mareike Thea; Hortschansky, Peter; Lopez-Berges, Manuel Sanchez; Heiss, Katharina; Beckmann, Nicola; Heigl, Thomas; Hermann, Martin; Krueger, Thomas; Kniemeyer, Olaf; Brakhage, Axel A.; Haas, Hubertus: The monothiol glutaredoxin GrxD is essential for sensing iron starvation in Aspergillus fumigatus. PLOS GENETICS. 2019; 15(9); e1008379.
- Furukawa, Takanori; Scheven, Mareike Thea; Misslinger, Matthia; Zhao, Can; Hoefgen, Sandra; Gsaller, Fabio; Lau, Jeffrey; Jöchl, Christoph; Donaldson, Ian; Valiante, Vito; Brakhage, Axel A; Bromley, Michael J;Haas, Hubertus; Hortschansky Peter.: The fungal CCAAT-binding complex and HapX display highly variable but evolutionary conserved synergetic promoter-specific DNA recognition. NUCLEIC ACID RES. 2020; 48(7); 3567-3590.
- Bauer, Ingo; Lechner, Lukas; Pidroni, Angelo; Petrone, Anna-Maria; Merschak, Petra; Lindner, Herbert; Kremser, Leopold; Graessle, Stefan; Golderer, Georg; Allipour, Shadab; Brosch, Gerald: Type I and II PRMTs regulate catabolic as well as detoxifying processes in Aspergillus nidulans. FUNGAL GENETICS AND BIOLOGY. 2019; 129(S); 86-100.
- Bauer, Ingo; Misslinger, Matthias; Shadkchan, Yana; Dietl, Anna-Maria; Petzer, Verena; Orasch, Thomas; Abt, Beate; Graessle, Stefan; Osherov, Nir; Haas, Hubertus: The Lysine Deacetylase RpdA Is Essential for Virulence in Aspergillus fumigatus. FRONTIERS IN MICROBIOLOGY. 2019; 10(S); 2773.
- Bauer, Ingo; Gross, Silke; Merschak, Petra; Kremser, Leopold; Karahoda, Betim; Sarikaya Bayram, Özlem; Abt, Beate; Binder, Ulrike; Gsaller, Fabio; Lindner, Herbert; Bayram, Özgür; Brosch, Gerald; Graessle, Stefan: RcLS2F - A Novel Fungal Class 1 KDAC Co-repressor Complex in Aspergillus nidulans.
FRONTIERS IN MICROBIOLOGY. 2020; 11; 43. - Huber, Anna; Oemer, Gregor; Malanovic, Nermina; Lohner, Karl; Kovács, Laura; Salvenmoser, Willi; Zschocke, Johannes; Keller, Markus A.; Marx, Florentine: Membrane sphingolipids regulate the fitness and antifungal protein susceptibility of Neurospora crassa. FRONTIERS IN MICROBIOLOGY. 2019; 10; 605.
- Holzknecht, Jeanett; Kühbacher, Alexander; Papp, Csaba; Farkas, Attila; Váradi, Györgyi; Marcos, Jose F.; Manzanares, Paloma; Tóth, Gábor K, Galgóczy, László; Marx, Florentine: The Penicillium chrysogenum Q176 antimicrobial protein PAFC effectively enhibits the growth of the opportunistic human pathogen Candida albicans. J FUNGI (Basel). 2020; 6(3); 141.
- Tóth, Liliána; Boros, Éva; Poór, Péter; Ördög, Attila; Kele, Zoltán; Váradi; Györgyi; Holzknecht, Jeanett; Bratschun-Khan, Doris; Nagy, István; Tóth, Gábor K.; Rákhely, Gábor; Marx, Florentine; Galgóczy, László: The potential use of the Penicillium chrysogenum antifungal protein PAF, the designed variant PAFoptand its γ-core peptide Pγopt in plant protection. MICROBIAL BIOTECHNOLOGY. 2020; 13(5); 1403-1414.
- Furukawa, Takanori; van Rhijn, Norman; Fraczek, Marcin; Gsaller, Fabio; Davies, Emma; Carr, Paul; Gago, Sara; Fortune-Grant, Rachael; Rahman, Sayema; Gilsenan, Jane M.; Houlder, Emma; Kowalski, Caitlin H.; Raj, Shriya; Paul, Sanjoy; Cook, Peter; Parker, Josie E.; Kelly, Steve; Cramer, Robert A.; Latge, Jean-Paul; Moye-Rowley, Scott; Bignell, Elaine; Bowyer, Paul; Bromley, Michael J: The negative cofactor 2 complex is a key regulator of drug resistance in Aspergillus fumigatus. NATURE COMMUNICATIONS. 2020; 11; 427
- Birstonas, Lukas; Dallemulle, Alex; Lopez-Berges, Manuel S.; Jacobsen, Ilse D.; Offterdinger, Martin; Abt, Beate; Strassburger, Maria; Bauer, Ingo; Schmidt, Oliver; Sarg, Bettina; Lindner, Herbert; Haas, Hubertus; Gsaller, Fabio: Multiplex Genetic Engineering Exploiting Pyrimidine Salvage Pathway-Based Endogenous Counterselectable Markers. MBIO. 2020; 11; e00230-20.
- Habeler, Matthias; Redl, Bernhard: Phage-display reveals interaction of lipocalin allergen Can f 1 with a peptide resembling the antigen binding region of a human γδT-cell receptor. BIOLOGICAL CHEMISTRY, 2020. Online ahead of print.
- Habeler, Matthias; Lindner, Herbert H.; Redl, Bernhard: A role of heparan sulphate proteoglycan in the cellular uptake of lipocalins ß-lactoglobulin and allergen Fel d 4. BIOLOGICAL CHEMISTRY 2020; 401(9); 1081-1092.
- Huang, Anming; Kremser, Leopold; Schuler, Fabian; Wilflingseder, Doris; Lindner, Herbert; Geley, Stephan; Lusser, Alexandra: Phosphorylation of Drosophila CENP-A on serine 20 regulates protein turn-over and centromere-specific loading. NUCLEIC ACIDS RESEARCH. 2019; 47(20); 10754-10770.
- Trixl, Lukas; Lusser, Alexandra: Getting a hold on cytosine methylation in mRNA. NATURE STRUCTURAL & MOLECULAR BIOLOGY, 2019; 26(5), 339–340.
- Gasser, Catherina; Delazer, Isabel;Neuner, Eva; Pascher, Katharina; Brillet, Karl; Klotz, Sarah; Trixl, Lukas; Himmelstoß, Max; Ennifar, Eric; Rieder*, Dietmar; Lusser*, Alexandra; Micura*, Ronald: Thioguanosine conversion enables mRNA life-time evaluation by RNA sequencing via double metabolic labeling. ANGEWANDTE CHEMIE INTERNATIONAL EDITION ENGL. 2020; 1–8. *co-corresponding authors
Selection of Funding
- Novel 5-flucytosine based antifungal therapeutics, Austrian Science Fund FWF P31093 (2018), Fabio Gsaller
- Iron regulation and virulence in Aspergillus fumigatus, Austrian Science Fund FWF M 2867 Meitner-Programm (2019), Clara Baldin,
- RNA-DECO: Decorating RNA for a Purpose, SFB FWF F8009-B Teilprojekt (2019), Alexandra Lusser
- The role of 5-methylcytosine in mammalian mRNA, Austrian Science Fund FWF P33936-B (2020), Alexandra Lusser
- SupErA: Siderophore-iron uptake in Erwinia amylovora and A. fumigatus” Euregio IPN 95-B30 (2020), Hubertus Haas
Collaborations
- Axel Brakhage, F. Schiller University Jena, Germany
- Mike Bromley, The Manchester University, UK
- Patrick Heun, University of Edinburgh, UK
- Nick D. Read, University of Manchester, UK; José F. Marcos, IATA, Valencia, Spain
- Franz Bracher, LMU Munich, Munich, Germany
- Laura Alcazar-Fuoli, Instituto de Salud Carlos III, Madrid, Spain
- Ilse Jacobsen, Hans-Knöll Institut, Jena, Germany
- Nermina Malanovic, University of Graz, Graz, Austria
- László Galgóczi, University of Szeged, Hungary
- Nir Osherov, Tel-Aviv University, Israel
 Univ.-Prof. Mag.Dr.rer.nat Hubertus Haas
Univ.-Prof. Mag.Dr.rer.nat Hubertus Haas
Director
Contact:
Innrain 80-82
6020 Innsbruck
Austria
Email: Hubertus.haas@i-med.ac.at
Phone: +43 512 9003 70205
Fax: +43 512 9003 73100
https://www.i-med.ac.at/molbio/



