Research
Cell Cycle and Cell Proliferation
at the Biocenter, Area Biochemistry and Chemistry, Innsbruck
Research Leader: Prof. Dr. Ludger Hengst PhD
|
Our group investigates molecular mechanisms that permit or restrict cell proliferation in mammalian cells. Deregulation of cell proliferation can lead to various human diseases including cancer. We therefore also study how these mechanisms are deregulated during oncogenesis.
Cells decide and commit to divide during a specific window in the mammalian cell cycle or during quiescence. At this time, they are especially responsive to various mitogenic and antimitogenic signals. We study how these diverse signals are integrated and how they impinge on the cell cycle control machinery. At the core of this machinery is a conserved family of protein kinases called cyclin-dependent kinases (Cdks). Cdk inhibitor proteins bind to these kinases and regulate their catalytic activity.
Current research projects in the lab focus on two main areas: Function and regulation of Cdk- inhibitory proteins and the role of translational control for the decision between cell proliferation and withdrawal from the cell cycle.
|
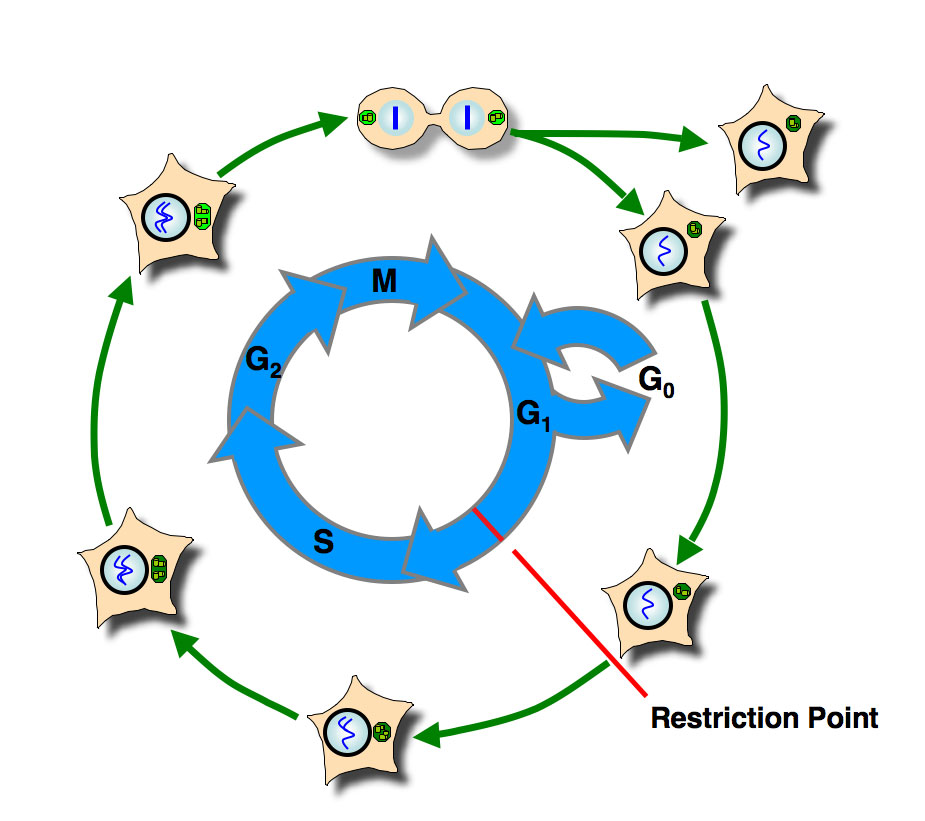
The eucaryotic cell division cycle is divided into four phases. DNA replication during S-phase is separated by so-called gap-phases, G1 and G2, from the segregation of the duplicated DNA and other cellular components in mitosis. At the end of M-phase, two daugher cells are generated by cytokinesis. Until they progress over the restriction point in G1-phase, cells frequently respond to growth or differentiation signals tat can lead to proliferation or growth arrest.
|
||
Med. Biochemistry – Research Group Prof. Hengst

Research Leader
Cell Cycle and Cell Proliferation
at the Biocenter, Area Biochemistry and Chemistry, Innsbruck
|
Independent Group Leader : Professor of Medical Biochemistry:
|
Research Group Phone: |

Research
Cell Cycle and Cell Proliferation
at the Biocenter, Area Biochemistry and Chemistry, Innsbruck
Research Leader: Prof. Dr. Ludger Hengst PhD
Research Group Staff
Administration Staff

Research – Open positions
Cell Cycle and Cell Proliferation
at the Biocenter, Area Biochemistry and Chemistry, Innsbruck
Research Leader: Dr. Ludger Hengst
Open positions:
We are interested in considering applications for:
- Postdoctoral fellows
- PhD students
- Diploma / Master students
Please contact Prof. Ludger Hengst (ludger.hengst@i-med.ac.at) further information.

Research
Cell Cycle and Cell Proliferation
at the Biocenter, Area Biochemistry and Chemistry, Innsbruck
Research Leader: Prof. Dr. Ludger Hengst PhD
Publications:
-
Grimmler, M., Wang, Y., Mund, T., Cilensek, Z., Keidel, E.M., Waddell, M.B., Jäckel, H., Kullmann, M., Kriwacki, R.W. and Hengst, L. (2007). The Cdk-inhibitory activity and stability of
p27Kip1 are directly regulated by oncogenic tyrosine kinases. Cell, 128, 269 – 280.
— -
Chu, I.M.; Sun, J., Arnaout, A, Kahn, H., Hanna, W., Narod, S., Sun, P., Keat-Tan, C.; Hengst, L. and Slingerland J. M. (2007). p27 phosphorylation by Src regulates inhibition of Cyclin E / Cdk2 and p27 proteolysis. Cell, 128, 281-294.
— -
Schmees, C, Prinz, C, Treptau, T., Rad, R., Hengst, L., Voland, P., Bauer, S., Brenner, L., Schmid, R. M. and Gerhard, M. (2007). Inhibition of T cell proliferation by Helicobacter pylori γ-glutamyl transpeptidase. Gastroenterology, 132, 1820 – 1833.
— -
Geley, S. and Hengst, L. (2008). Zellzyklus und Spindelapparat. In: Die Onkologie. 2. Auflage. W. Hiddemann, Bartram C. R. (Eds.). Springer Verlag. In press.
— -
Halfter, H., Friedrich, M., Resch, A., Kullmann, M., Stögbauer, F., Ringelstein, E.B. and Hengst, L. (2006). Oncostatin M induces growth arrest by inhibition of Skp2, Cks1, and cyclin A expression and induced p21 expression. Cancer Research 66, 6530-6539.
— -
Hengst, L. and Nigg, E.A. (2006). Cell cycle – An overview. Encyclopedic Reference of Genomics and Proteomics in Molecular Medicine. D. Ganten und K. Ruckpaul (Eds.). Springer Verlag 2006.
— -
Jaschke, B., Michaelis, C., Milz, S., Vogeser, M., Mund, T., Hengst, L., Kastrati, A., Schömig, A., Wessely, R. (2005). Local statin therapy differentially interferes with smooth muscle and endothelial cell proliferation and reduces neointima on a drug-eluting stent platform. Cardiovasc Res. 68, 483-492.
— -
Gerhard, M., Schmees, C., Voland, P., Endres, N., , Sander,M., Reindl, W., Rad, R., Oelsner, M., Decker,T., Mempel, M., Hengst, L. and Prinz, C. (2005). A secreted low molecular weight protein from Helicobacter pylori induces cell cycle arrest of T-cells. Gastroenterology 128, 1327-1339.
— -
Swaminathan, S., Körner, R., Kiendl, F., Luptti, R., Hengst, L. and Melchior, F. (2004). RanGAP*SUMO-1 is phosphorylated at the onset of mitosis and remains associated with RanBP2 upon NPC disassembly. Journal of Cell Biology 164, 965-971.
— -
Lacy, E.R., Filippov, I., Otieno, S., Xiao, L., Lewis, W.S., Sivakolundu, S., Weiss, S., Hengst, L. and Kriwacki, R.W. (2004). p27 Binds Cyclin/Cyclin-dependent Kinase Complexes via a Sequential Mechanism involving Binding-induced Protein Folding. Nature Structural & Molecular Biology 11, 358-364.
— -
Hengst, L. (2004). A second RING to destroy p27Kip1. Nature Cell Biology, 6, 1153-1155.
— -
Pendergraft III, W.F., Rudolph, E.H., Grimmler, M., Hengst, L., Falk,R.J., Jennette, J.C. and Preston, G. A. . (2004). Proteinase 3 sidesteps caspases and cleaves p21Waf1/Cip1/Sdi to induce endothelial cell apoptosis. Kidney International 65, 75-84.
— -
Göpfert, U. , Kullmann, M. and Hengst, L (2003). Cell Cycle dependent translation of p27 involves a responsive element in its 5’UTR that overlaps with an uORF. Human Molecular Genetics 12. 1767-1779.
— -
Connor, M.K., Kotchetkov, R., Cariou, S., Beniston, R.G., Resch, A., Lupetti, R., Melchior, F., Hengst, L.*, and Slingerland, J.M.* (2003). *Communicating authors.CRM1/Ran-mediated nuclear export of p27Kip1 involves a nuclear export signal and links p27 export and proteolysis. Molecular Biology of the Cell 14. 201-213.
— -
Wessely, R., Hengst, L., Wegener, F., Richter, T., Paskalidis, M., Schomig, A., Brandl, R., Neumann, F.J.(2003). IRF-1 is essential for the limitation of neointima hyperplasia: Implications for the pathophysiology of neointima formation and therapeutic prevention of restenotic lesions following coronary angioplasty. Human Molecular Genetics, 12. 177-187.
— -
Kullmann M., Göpfert, U., Siewe, B. and Hengst, L. (2002). ELAV/Hu proteins inhibit p27 translation through an IRES element in the p27 5’UTR. Genes & Development 16, 3087-3099.
— -
Melchior, F. and Hengst, L. (2002). Sumo1-p53. Cell Cycle 1, 245-249.
— -
Melchior, F and Hengst, L. (2000). Mdm2-SUMO1: Is bigger better? Nature Cell Biology 2, E161-E163.
— -
Hengst, L., Göpfert, U., Lashuel, H. A. and Reed, S.I. (1998). Complete inhibition of Cdk/Cyclin by one molecule of p21Cip1. Genes & Development 12, 3882-3888.
— -
Hengst, L. and Reed, S.I. (1998). Inhibitors of the Cip/Kip Family. Current Topics in Microbiology and Immunology, 227. 25-41
— -
Niculescu III, A., Chen, X., Smeets, M., Hengst, L., Prives, C. and Reed, S. I. (1998). Effects of p21Cip1/Waf1 on both the G1/S and the G2/M cell cycle transitions: pRb as a critical determinant in blocking and in preventing endoreduplication. Mol. Cell. Biol., 18. 629-643.
— -
Kiwacki, R.W., Hengst, L., Tennant, L., Reed, S.I. and Wright, P. E. (1996). Structural studies of p21Waf1/Cip1/Sdi1 in the free and Cdk2 bound state: conformational disorder mediates binding diversity. PNAS, 93. 11504-11509.
— -
Hengst, L. and Reed, S.I. (1996). Translational control of p27Kip1 accumulation during the cell cycle. Science, 271. 1861-1864.
— -
Hengst, L., Grabowski, R. and Gallwitz, D.(1996). Ypt6. In: Guidebook to the small GTPases. M. Zerial und L.A. Huber. Oxford University Press.
— -
Hengst, L. and Gallwitz, D. (1996). Ryh1. In: Guidebook to the small GTPases. M. Zerial und L.A. Huber. Oxford University Press.
— -
Resnitzky, D., Hengst, L. and Reed, S.I.R. (1995). Cyclin A associated kinase activity is rate-limiting for entrance into S phase, and is negatively regulated in G1 by p27Kip1. Mol Cell Biol., 15 . 4347-4352.
— -
Reed, S.I., Bailly, E., Dulic, V., Hengst, L., Resnitzky, D., and Slingerland, J. (1994). G1 control in mammalian cells. J. Cell Sci. – Suppl.18: 69-73.
— -
Slingerland, J.M., Hengst, L., Alexander, D., Stampfer, M.R. and Reed, S.I. (1994). A novel inhibitor of Cyclin/Cdk activity detected in transforming growth factor -arrested epithelial cells. Mol. Cell. Biol., 14: 3683-3694.
— -
Hengst, L., Dulic, V,. Slingerland, JM., Lees, E. and Reed SI. (1994). A cell cycle-regulated inhibitor of cyclin-dependent kinases. PNAS, 91. 5291-5295.
— -
Kibbe, W., Hengst, L. and Gallwitz, D. (1993). The Ypt Gene family in yeast. In: The ras superfamily of GTPases. Edited by F. McCormick and J.C. Lacal. CRC Press Inc.
— -
Bednarek S. Y., Reynolds T. L., Schroeder M., Grabowski R., Hengst L., Gallwitz D. and Raikhel, N.V. (1994). A small GTP-binding protein from Arabidopsis thaliana functionally complements the yeast YPT6 null mutant. Plant Physiol. 104 : 591-596
— -
Wichmann, H., Hengst, L., Gallwitz, D., Schimmoeller, F. and Riezman, H. (1994). YPT7. In: Rothblatt, J., Novick, P., Stevens, T.H.: Guidebook to the secretory pathway. 272 Oxford University Press.
— -
Wichmann, H., Hengst, L. and Gallwitz, D. (1992). Endocytosis in yeast: Evidence for the involvement of a small GTP-binding protein (Ypt7p). Cell 71, 1131-1142.
— -
Wagner, P., Hengst, L. and Gallwitz, D. (1992). Ypt proteins in yeast. Methods Enzymol., 219, 369-387.
— -
Gallwitz, D., Becker, J., Benli, M., Hengst, L., Morsin-Huaman, C., Tan, J., Vollmer, P. and Wichman, H. (1991). The YPT-branch of the ras superfamily in yeast: functional importance of the putative effector region. In: NATO ASI-Series, Plenum press, New York.
— -
Hengst, L., Lehmeier, T. and Gallwitz, D. (1990). The ryh1 gene in the fission yeast Schizosaccharomyces pombe encoding a GTP-binding protein related to ras, rho and ypt: structure, expression and identification of its human homologue. EMBO J., 9, 1949-1955.
— -
Kadenbach, B., Schlerf, A., Mengel, T., Hengst, L., Cao, X., Suske, G., Eckerskorn, C. and Lottspeich, F. (1989). Tissue-spezific expression of nuclear genes for mitochondrial enzymes. In: Organelles in eucaryotic cells. (eds: Tager, J.M., Azzi, A., Papa, S. and Guerrieri, F.). Plenum press, New York.
— -
Cao, X., Hengst, L., Schlerf, A., Droste, M., Mengel, T. and Kadenbach, B. (1988). Complexity of nuclear-encoded genes of mammalian cytochrom c oxidase. Ann. N.Y. Acad. Sci., 550, 337-347.
LERNUNTERLAGEN